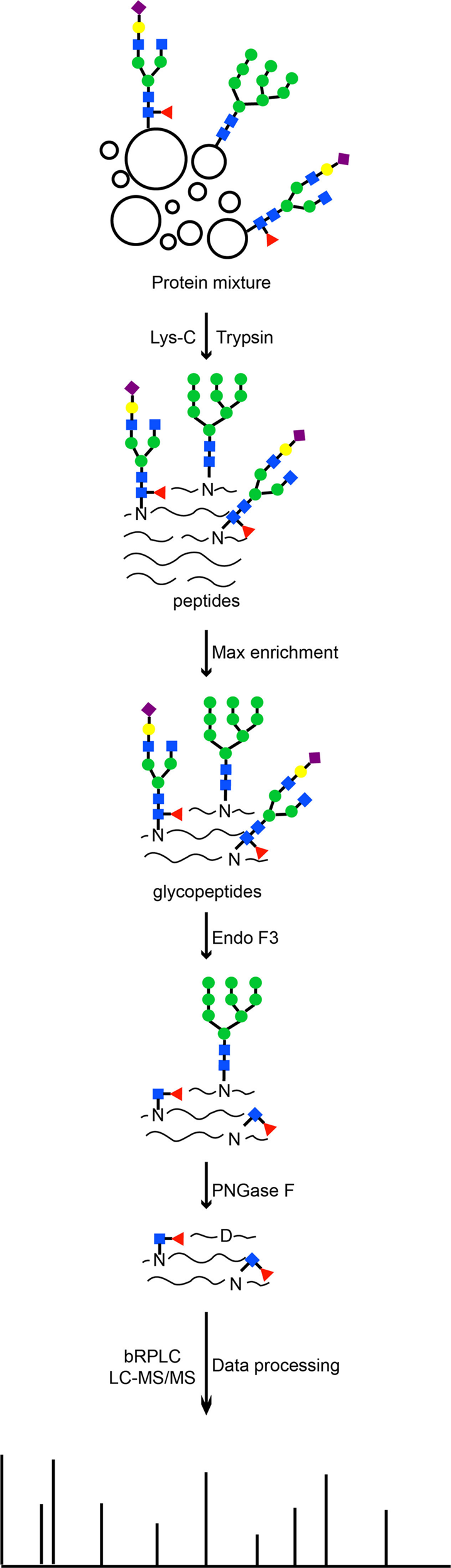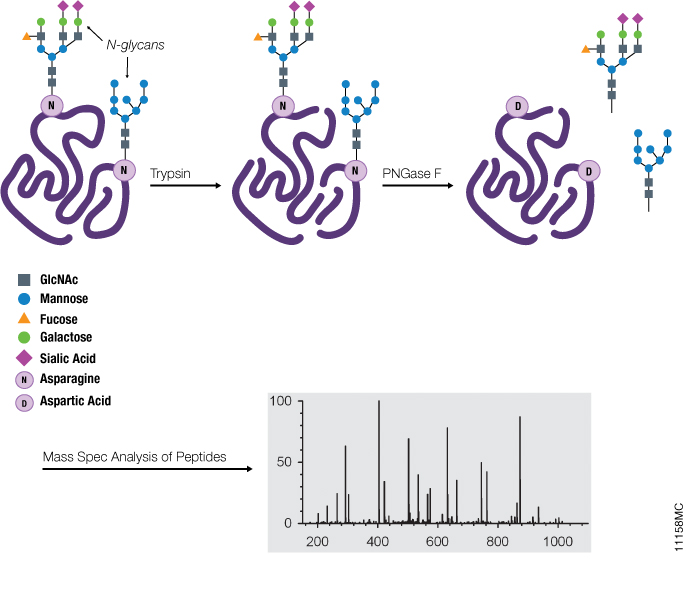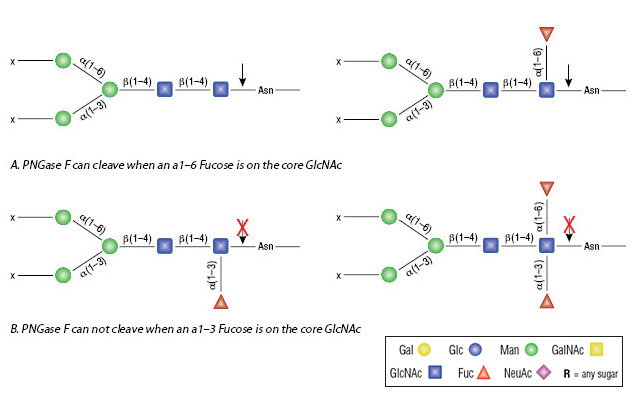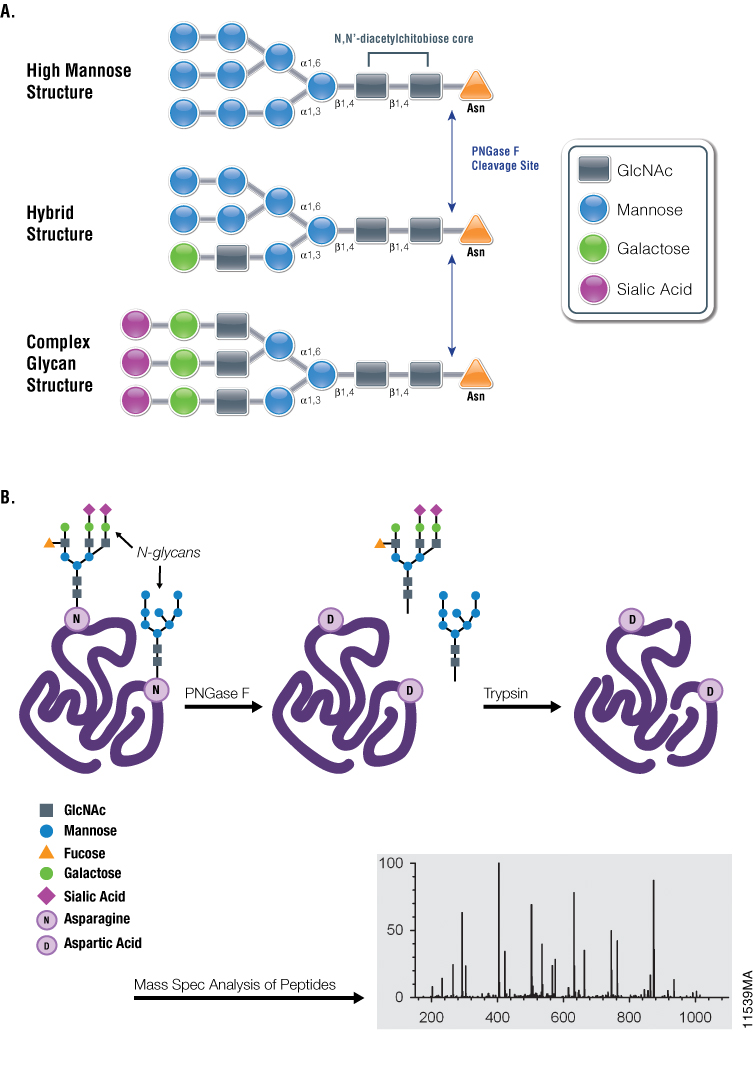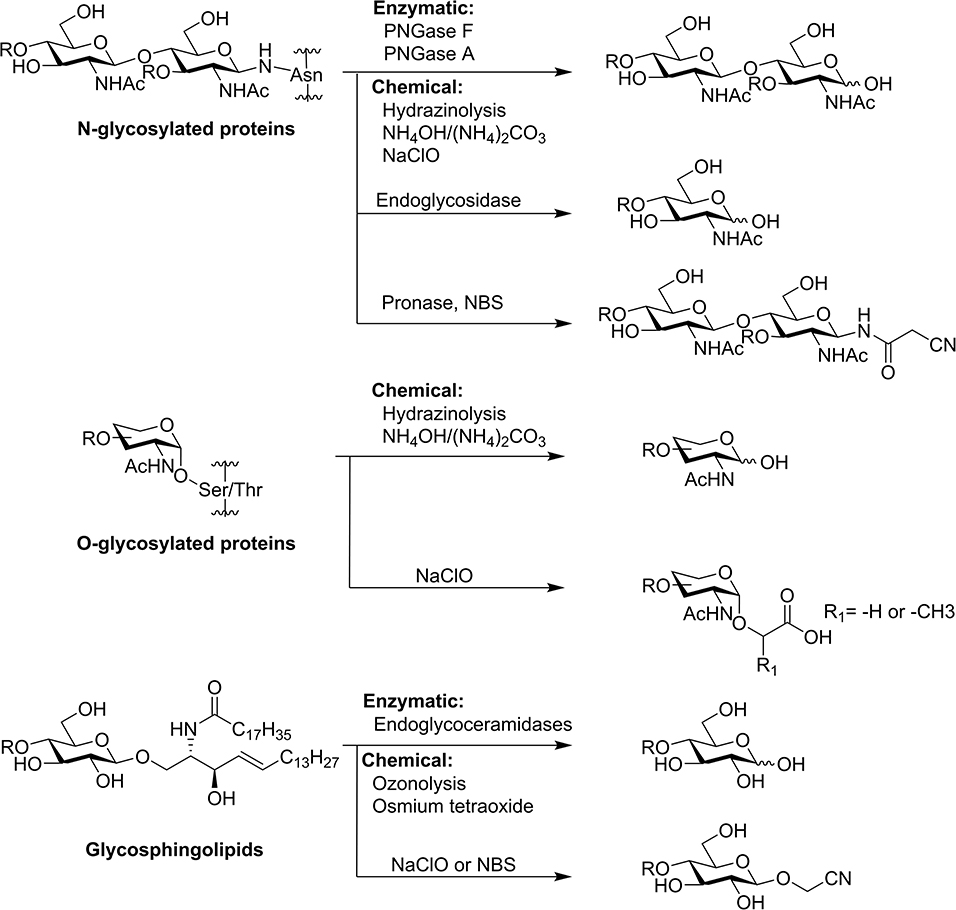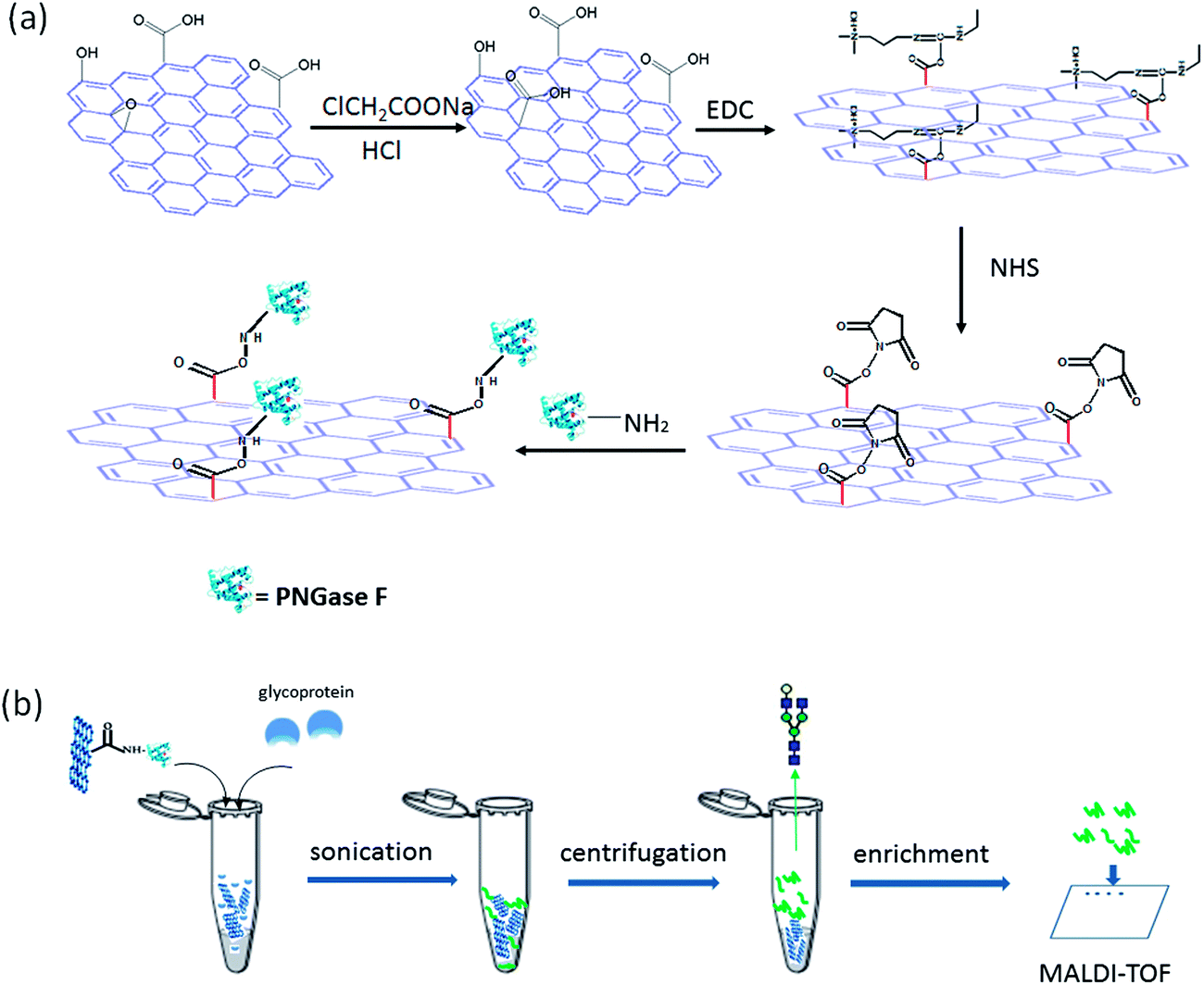
A graphene oxide-based immobilized PNGase F reagent for highly efficient N -glycan release and MALDI-TOF MS profiling - Analytical Methods (RSC Publishing) DOI:10.1039/C3AY41829D
A graphene oxide-based immobilized PNGase F reagent for highly efficient N-glycan release and MALDI-TOF MS profiling - Analytical Methods (RSC Publishing)

A Novel PNGase Rc for Improved Protein N-Deglycosylation in Bioanalytics and Hydrogen–Deuterium Exchange Coupled With Mass Spectrometry Epitope Mapping under Challenging Conditions | Analytical Chemistry
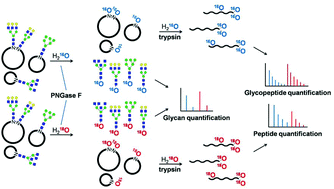
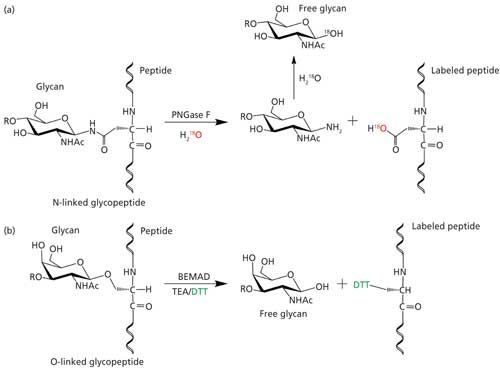
PNGase F-mediated incorporation of 18O into glycans for relative glycan quantitation - Analyst (RSC Publishing)

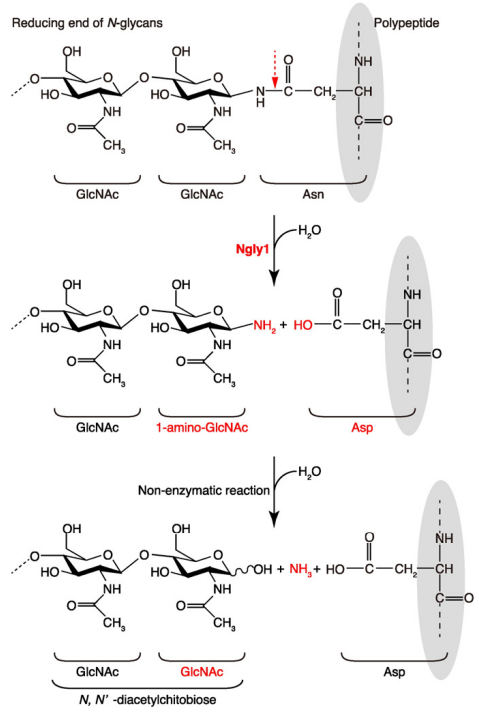
Proposed mechanism of deglycosylating enzyme PNGase and design of its... | Download Scientific Diagram
![PDF] Discovery and characterization of a novel extremely acidic bacterial N-glycanase with combined advantages of PNGase F and A | Semantic Scholar PDF] Discovery and characterization of a novel extremely acidic bacterial N-glycanase with combined advantages of PNGase F and A | Semantic Scholar](https://d3i71xaburhd42.cloudfront.net/c11ed2f93d2f81794dcbc0a37cf17ffb815bc5f4/2-Figure1-1.png)
PDF] Discovery and characterization of a novel extremely acidic bacterial N-glycanase with combined advantages of PNGase F and A | Semantic Scholar

Deciphering Protein O-Glycosylation: Solid-Phase Chemoenzymatic Cleavage and Enrichment | Analytical Chemistry

Synthesis of PNGase-resistant N-glycopeptide containing an α-anomeric glycosidic linkage: Journal of Carbohydrate Chemistry: Vol 40, No 7-9

Releasing N-glycan from Peptide N-terminus by N-terminal Succinylation Assisted Enzymatic Deglycosylation | Scientific Reports
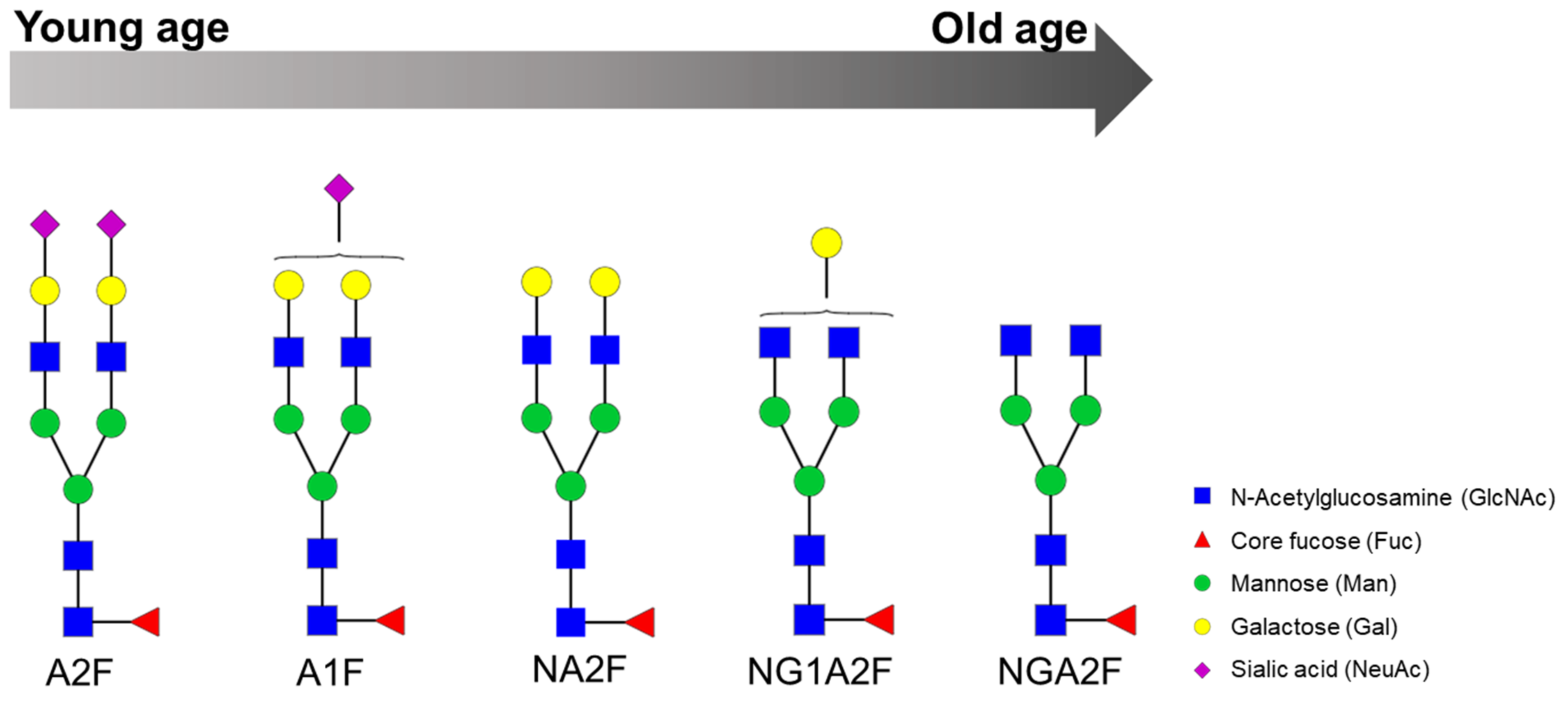
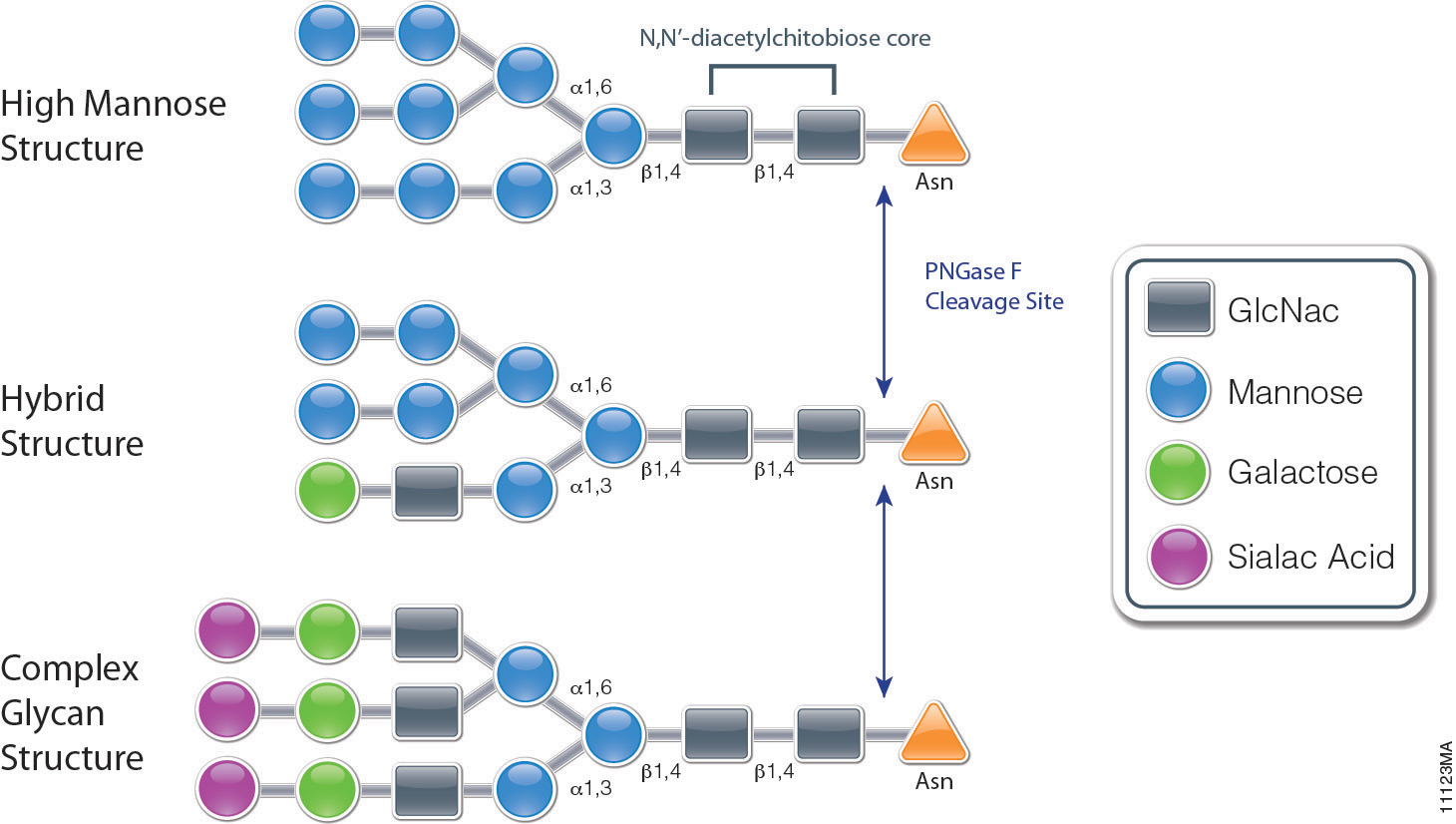
IJMS | Free Full-Text | Glycosylation Biomarkers Associated with Age-Related Diseases and Current Methods for Glycan Analysis