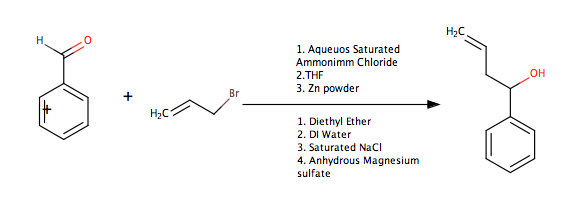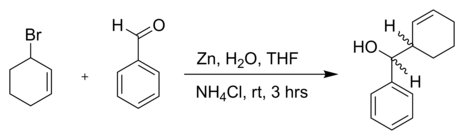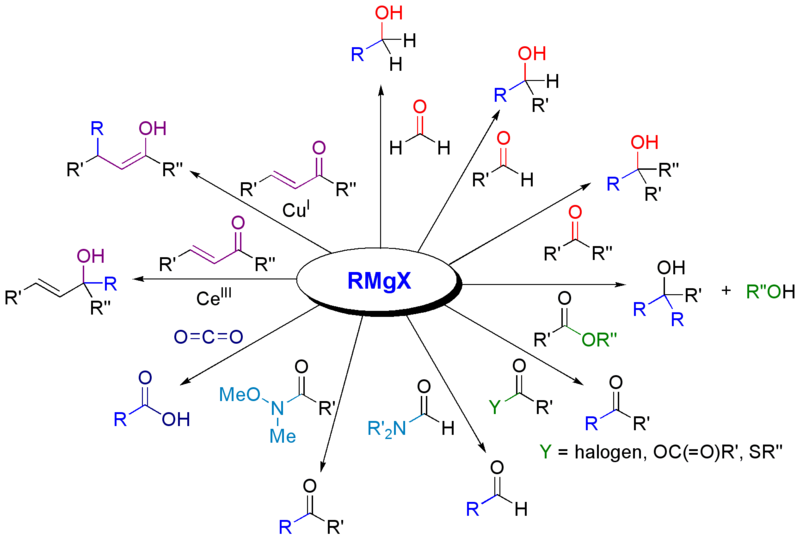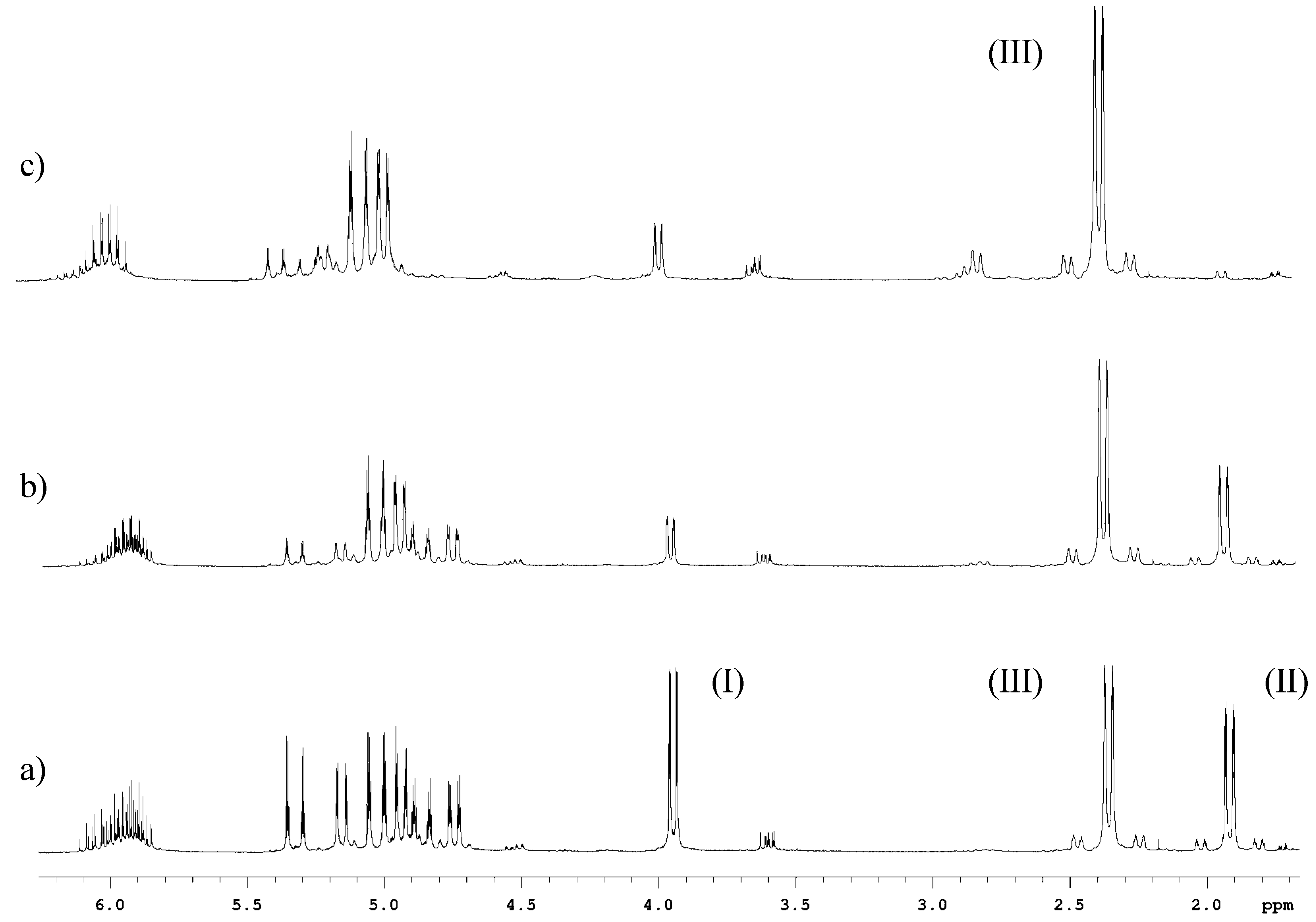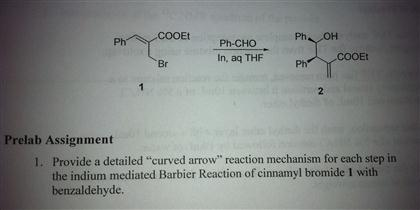
High regio- and stereoselective Barbier reaction of carbonyl compounds mediated by NaBF4/Zn (Sn) in water - New Journal of Chemistry (RSC Publishing)

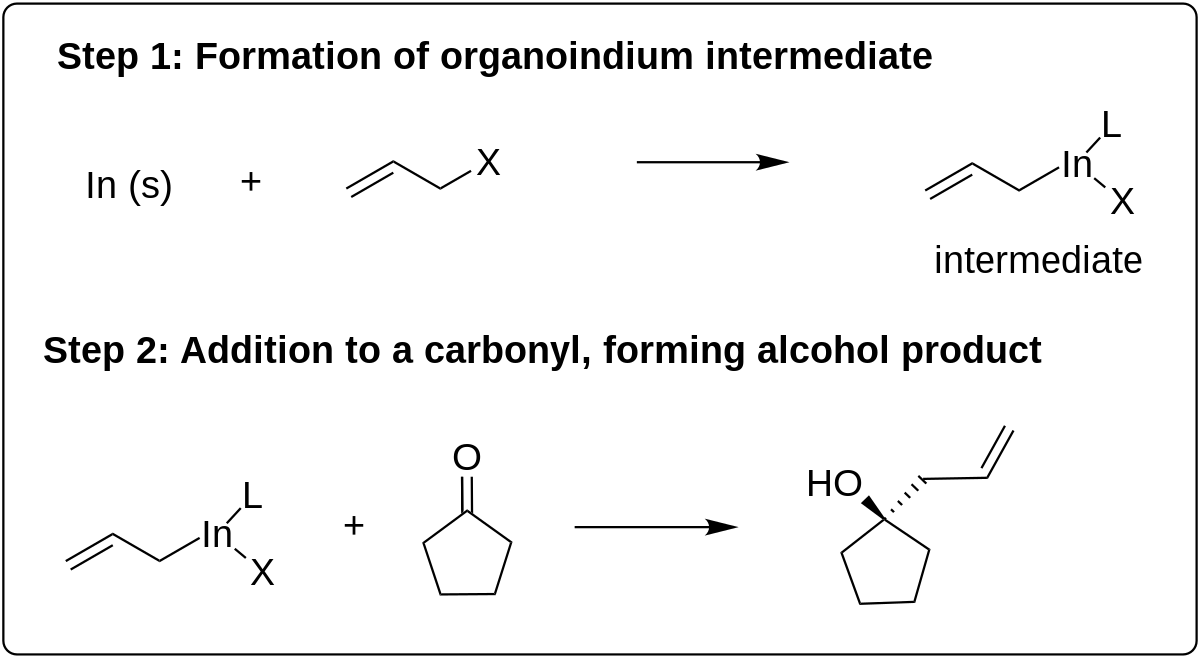
Proposed mechanism for the samarium Barbier reaction with excess HMPA. | Download Scientific Diagram
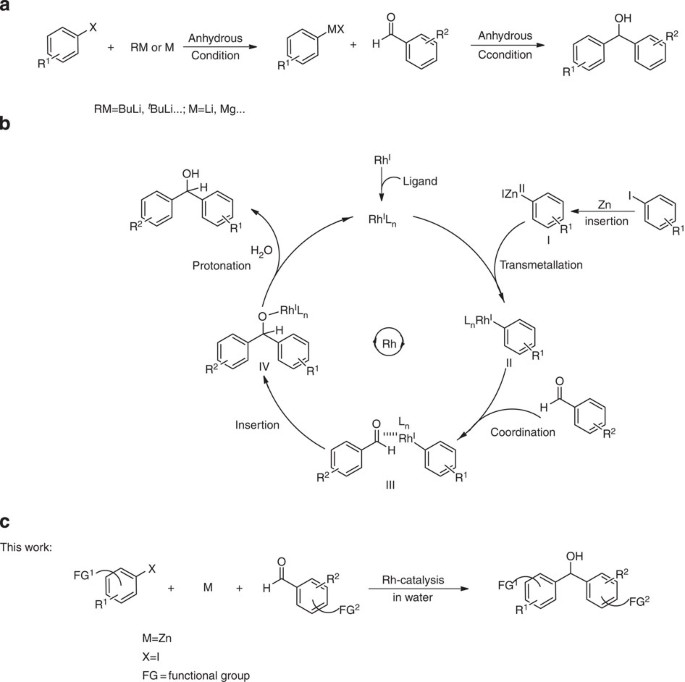
The Barbier–Grignard-type arylation of aldehydes using unactivated aryl iodides in water | Nature Communications

Preparation and Use of Samarium Diiodide (SmI2) in Organic Synthesis: The Mechanistic Role of HMPA and Ni(II) Salts in the Samarium Barbier Reaction | Protocol
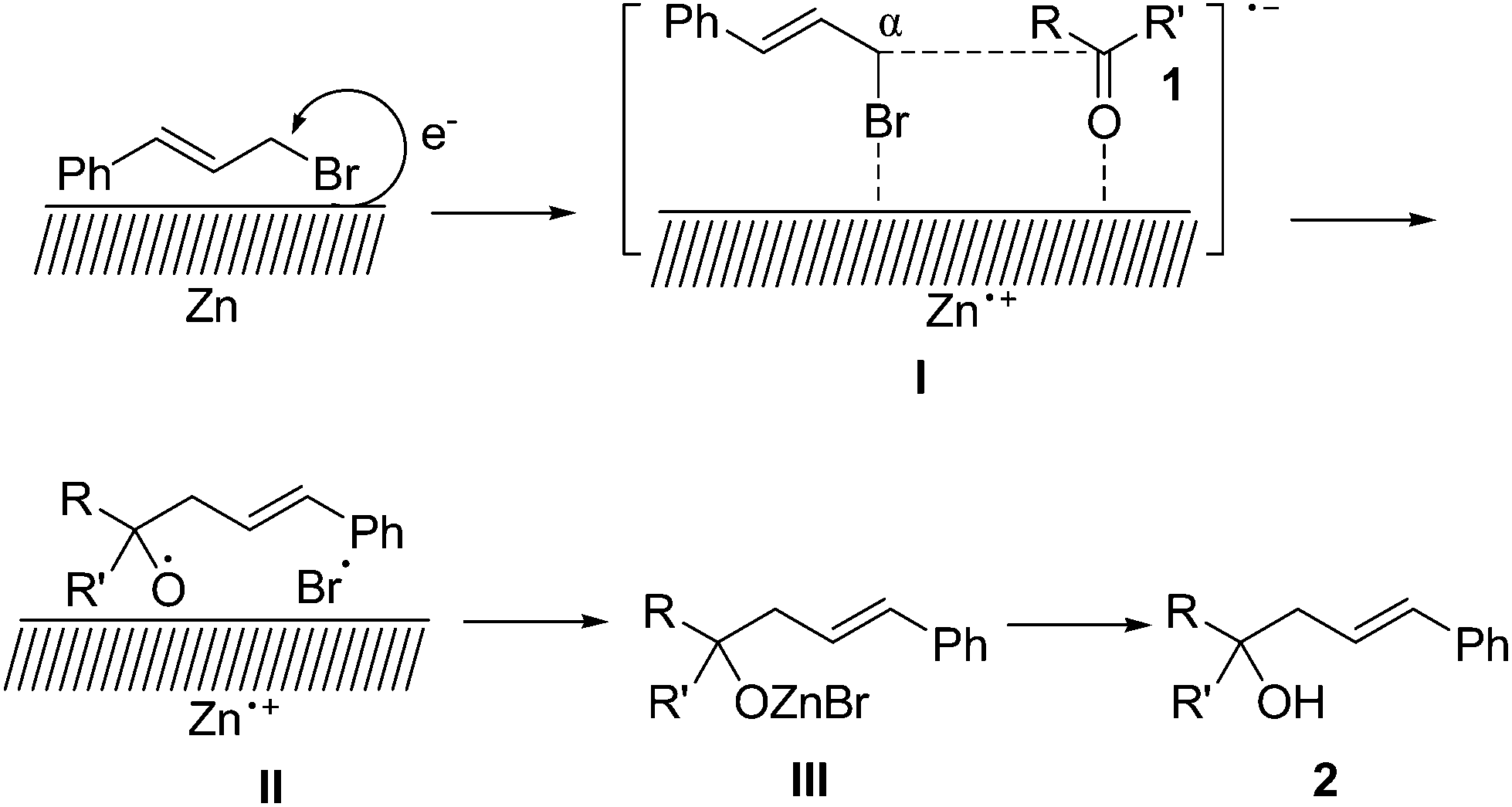
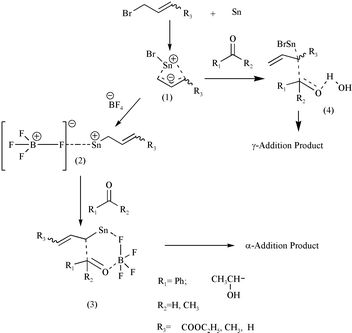
Zinc-mediated α-regioselective Barbier-type cinnamylation reactions of aldehydes, ketones and esters - Organic & Biomolecular Chemistry (RSC Publishing) DOI:10.1039/C7OB00723J
![bmim][Br] as an Inexpensive and Efficient Medium for the Barbier‐Type Allylation Reaction Using a Catalytic Amount of Indium: Mechanistic Studies - Dey - 2018 - European Journal of Organic Chemistry - Wiley Online Library bmim][Br] as an Inexpensive and Efficient Medium for the Barbier‐Type Allylation Reaction Using a Catalytic Amount of Indium: Mechanistic Studies - Dey - 2018 - European Journal of Organic Chemistry - Wiley Online Library](https://chemistry-europe.onlinelibrary.wiley.com/cms/asset/dfe491a2-a6b4-4b18-8779-e0cd12e8ee19/ejoc201800043-toc-0001-m.png)